As an industry leading clinical research solutions provider, OpenClinica’s electronic data capture (EDC) platform takes clinical data management to a whole new level. We automate electronic data capture (EDC) with dynamic, comprehensive electronic case report forms (eCRFs) for clinical trials, enabling researchers to collect better data, faster. Build, capture, and report on your clinical data with ease thanks to the power of OpenClinica.
Build
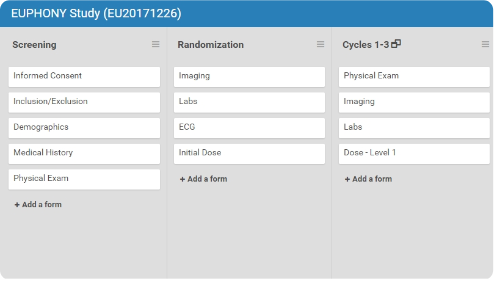
- Define, order, and re-order your events and forms on a drag-and-drop interface.
- Collaborate with your team on that same interface. Audit logs ensure that no changes are lost.
- Update your forms as needed: the study build system will track versioning for you.
- Preview the forms exactly how they’ll appear and function for your users from within the build system.
- Publish to a test or production environment in less than a minute.
- Invite study managers and sites with permissions you control.
- Create a site or user once, and the system stores the record for use in other studies.

Capture
- Make data entry a joy with flexible, mobile-friendly forms with real-time edit checks and auto-save.
- Upload data in batches with simple imports or via an extensive web services API that connects to your existing systems.
- Collect data directly from participants conveniently from any device.
- Randomize seamlessly from within eCRFs.
- Export data in common file formats.
Report
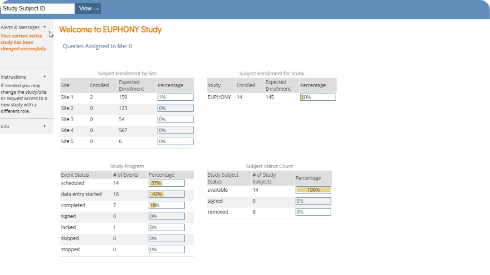
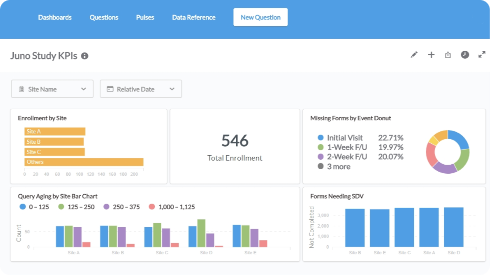
- Instantly access operational and clinical data to act quickly on everything from queries to adverse events.
- Visualize with bar charts, line graphs, and more.
- Send report updates to team leads and sites on a time- or event-based schedule.
Ready to see how OpenClinica makes the complex easy?

Sites
Keep your sites productive and compliant.Give sites an easy, and intuitive way to provide accurate study data. Auto-notify site users with actions required. Auto-manage study calendars. Apply electronic signatures.
Data Managers
Maintain control over your study data.Build, preview, publish, and repeat on a collaborative, drag-and-drop data visualization interface. Provision sites and users, resolve queries, and export datasets in a flash.
Monitors
Simplify your monitor's work.Give your monitors a mobile friendly dashboard to assist them in the field, showing what they need to do, and when they need to do it. Review site activities, verify data against source.
Study Participants
Get frictionless data capture from patients.Allow patients to provide data anytime, anywhere from their own device. Provide timely notifications when data is required.

Sponsors
Expedite your trial while maintaining data quality.Keep up-to-date with study progress. Save time and money. Maintain regulatory compliance. Rest well knowing your data is secure.
-
Optimized Cloud
Maximize the performance and reliability of your studies with stellar support, zero data loss, and disaster recovery. Rest assured with our validated and secure global infrastructure. SOC, ISO27001 certified.
-
Training and Support
Comprehensive hands-on training. Learn to build and manage your studies. Rapid support from OpenClinica experts.
-
Regulatory Compliance
Complies with GCP, 21 CFR Part 11, and other regulations. Complete validation documentation and audit support.
-
Cost Effective
Make better use of limited resources. With our EDC system, you can start small and scale as needed. OpenClinica is cost effective for any organization.
Resources
Check out our free tools and thought leadership to learn more about clinical data management, electronic data capture, ePRO/eCOA, decentralized clinical research, and more!







