In the previous post, we presented a cross-form example of clinical data collected in one event factoring into the normal lab range for a subsequent event. But clinical data aren’t the only factors that drive decisions. When an event occurred may determine when it should happen next. Dosing visits provide a common example. Depending on the protocol, dosing might occur at precise intervals (e.g. exactly 21 days between doses) or within windows (e.g. at least 7 days and no more than 10 days from the previous dose). Your EDC system should be able to enforce either type of scheduling, by reading not only the dates entered into forms, but dates found in form and event metadata.
In the example illustrated below, the form makes calculations between the start of a current event (“Dosing Visit 2”) and the start of the previous visit (“Dosing Visit 1”). According to this imaginary protocol, no fewer than 7 and no more than 10 days may elapse between these two visits.
- If dosing visit 2 occurs within this range, the form guides the site-based user on how to prepare the dose.
- If dosing visit 2 has a start date fewer than 7 days after dosing visit 1, the form displays instructions not to proceed, and provides the earliest and latest start dates for the visit.
- Finally, if dosing visit 2 has a start date greater than 10 days after dosing visit 1, the form displays instructions to submit a protocol deviation note.
All of these calculations and feedback take place instantaneously.
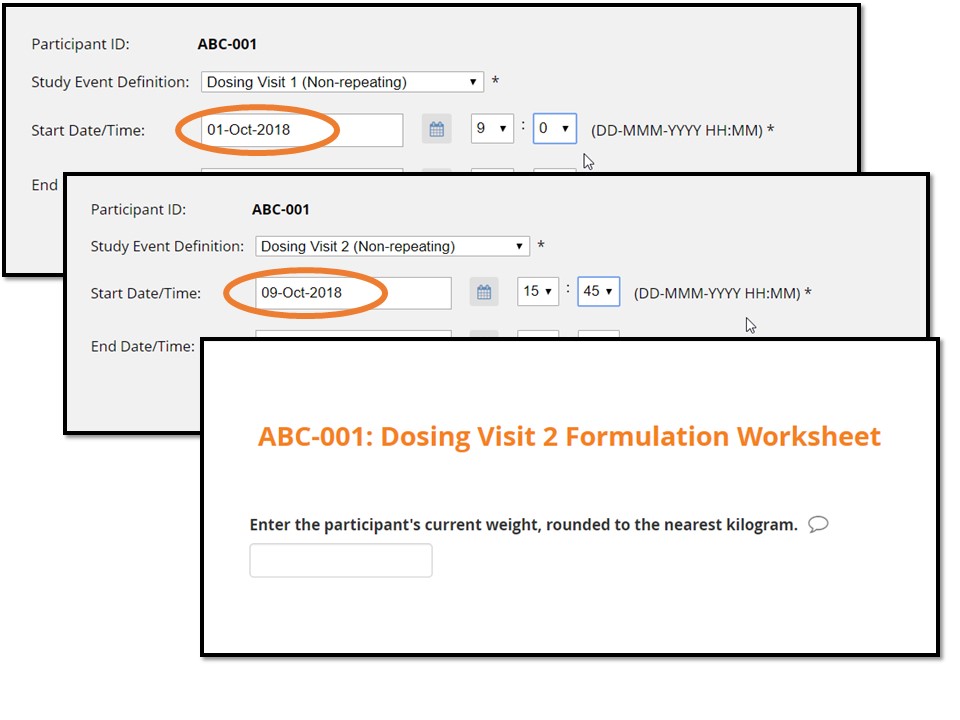
For this participant, Dosing Visit 2 is within window.
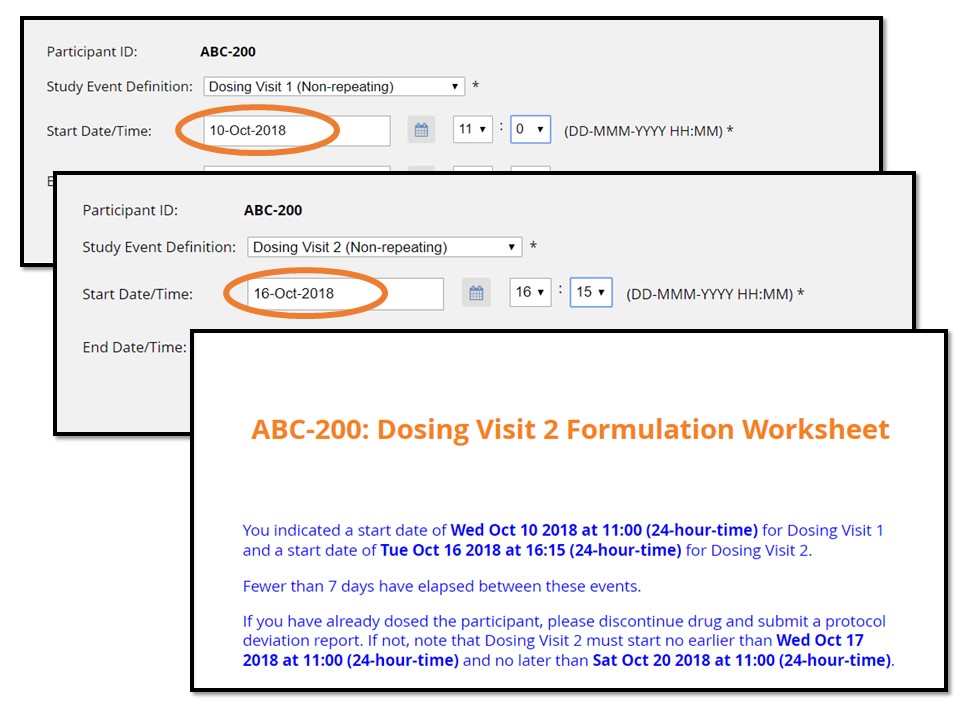
For this participant, Dosing Visit 2 has been scheduled for too early a date.
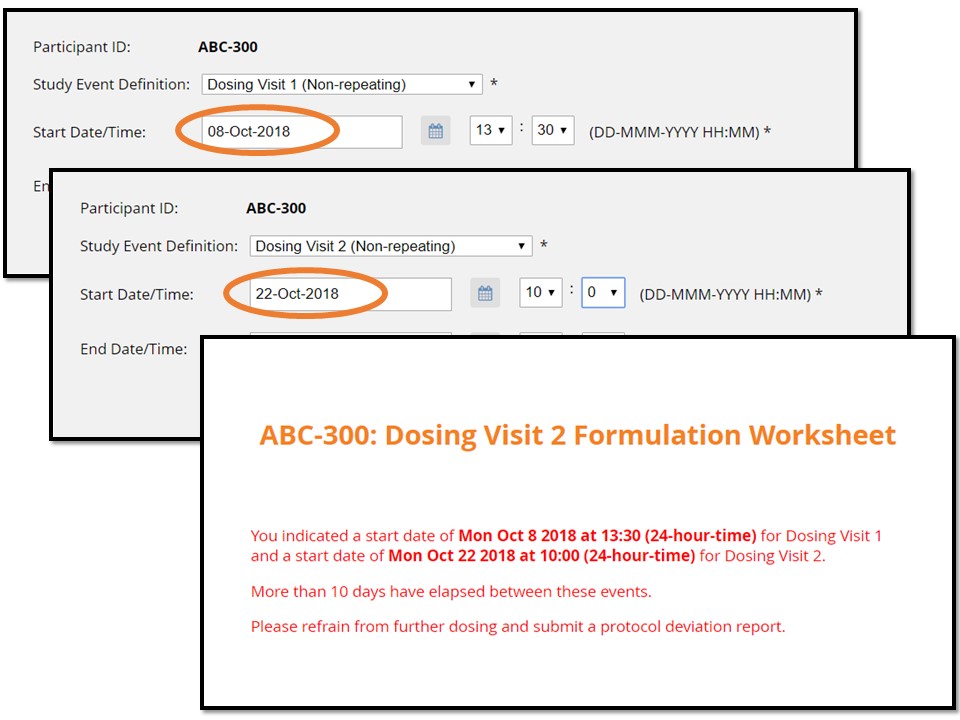
For this participant, Dosing Visit 2, were it to occur, would happen beyond the 10 day maximum from Dosing Visit 1.
Watch this webinar for more information on cross forms and the timing of clinical data capture!
