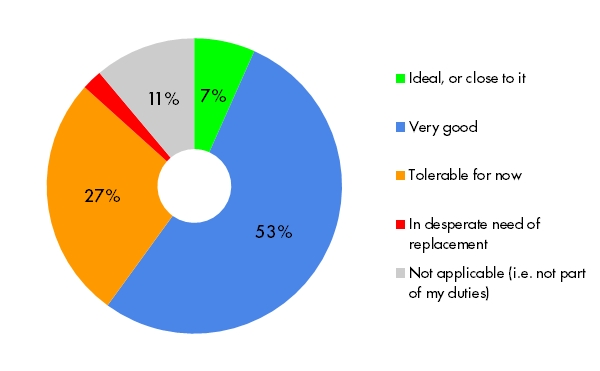
For the last several months, we’ve asked professionals with data management responsibilities to evaluate their current processes for accomplishing critical tasks. We then asked them to rank three of these tasks as their first, second, and third priorities.
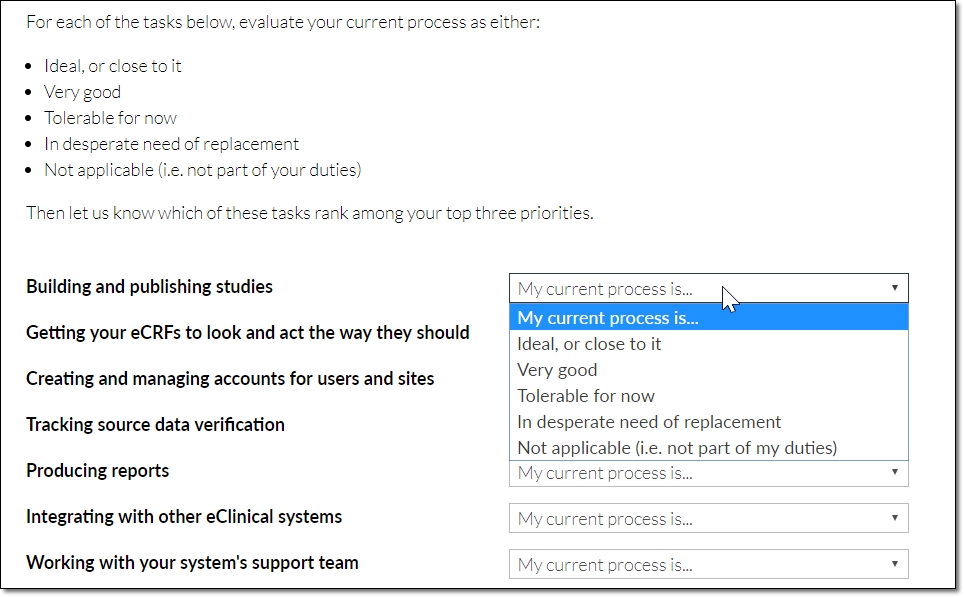
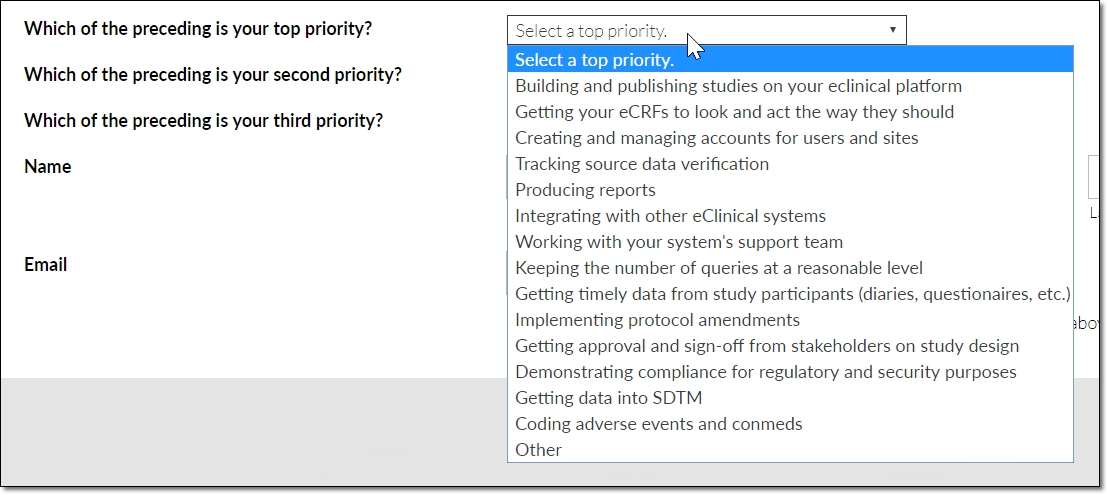
Specifically, we each respondent to consider these EDC-related tasks…
- Getting your eCRFs to look and act the way they should
- Producing reports
- Building and publishing studies on your eclinical platform
- Keeping the number of queries at a reasonable level
- Tracking source data verification
- Getting timely data from study participants (diaries, questionaires, etc.)
- Coding adverse events and conmeds
- Integrating with other eClinical systems
- Demonstrating compliance for regulatory and security purposes
- Implementing protocol amendments
- Getting data into SDTM
- Creating and managing accounts for users and sites
- Getting approval and sign-off from stakeholders on study design
- Working with your system’s support team
… and to characterize their process for each one as either…
- Ideal, or close to it
- Very good
- Tolerable for now
- In desperate need of replacement
- Not applicable (i.e. not part of my duties)
The data below reflects the responses of data managers (n=25), study and program leads ( n=13), IT/database professionals (n=6), and a CRA. To reduce bias, we did not include responses of known OpenClinica users.
What did we learn? Data managers and their colleagues are getting the job done (no surprise there) and are generally content with the way they’re doing it. But current processes usually fall short of ideal. How do your experiences compare?
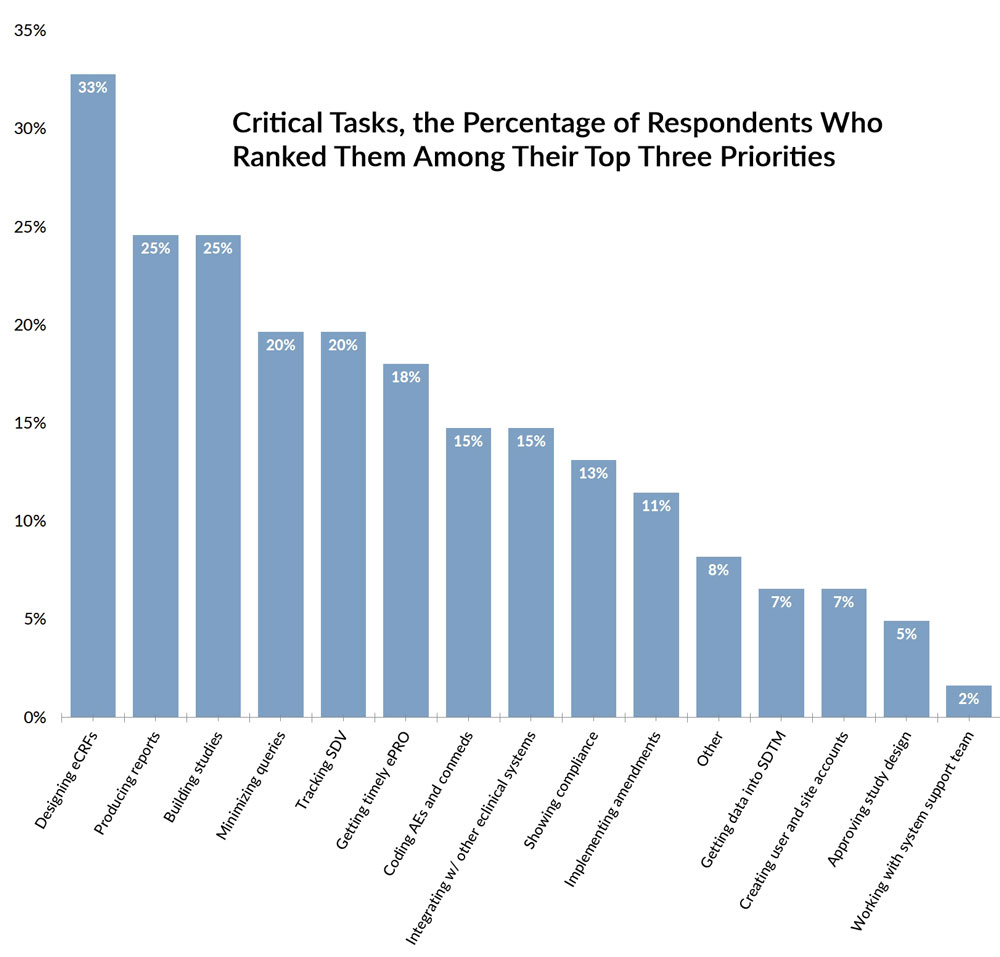
Click to see enlarge version
Getting eCRFs to look and act the way they should
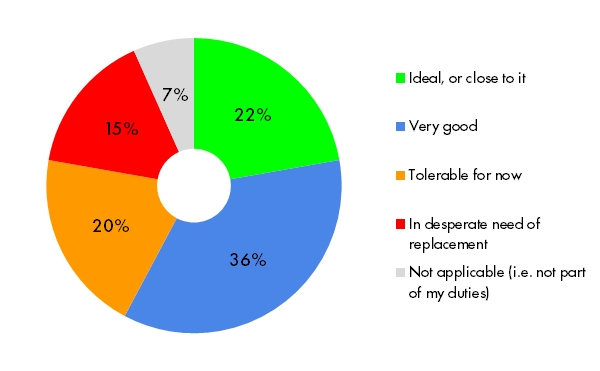
Producing reports
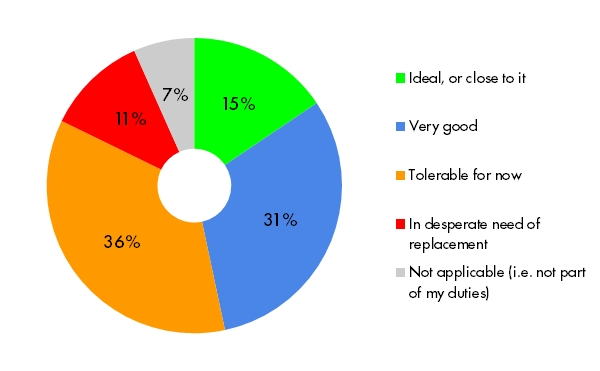
Building and publishing studies
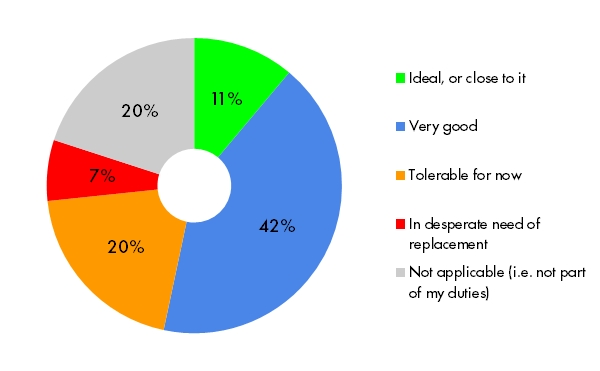
Keeping the number of queries at a reasonable level
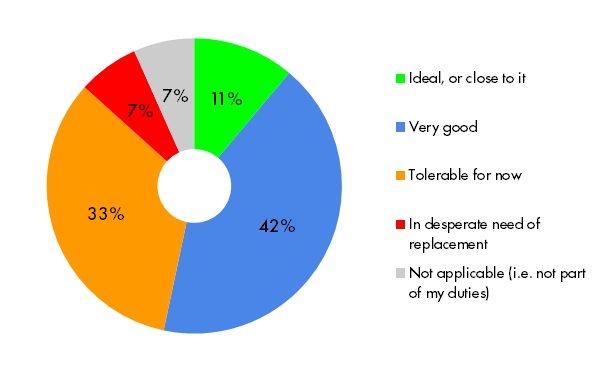
Tracking source data verification
![]()
Getting timely data from study participants (diaries, questionaires, etc.)
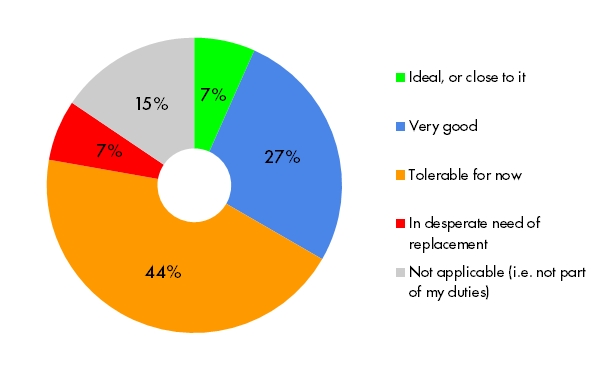
Coding adverse events and conmeds
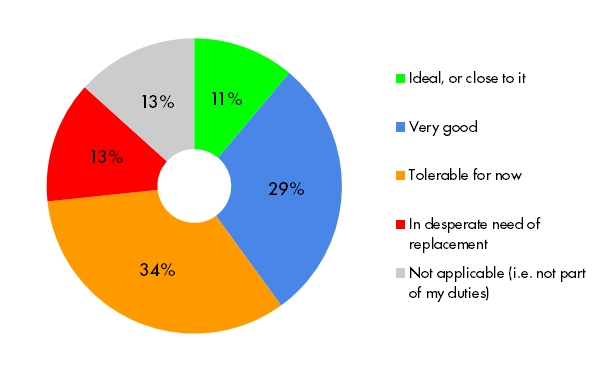
Integrating with other eClinical systems
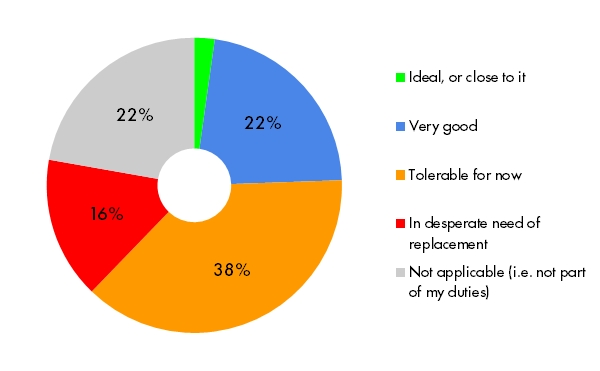
Demonstrating compliance for regulatory and security purposes
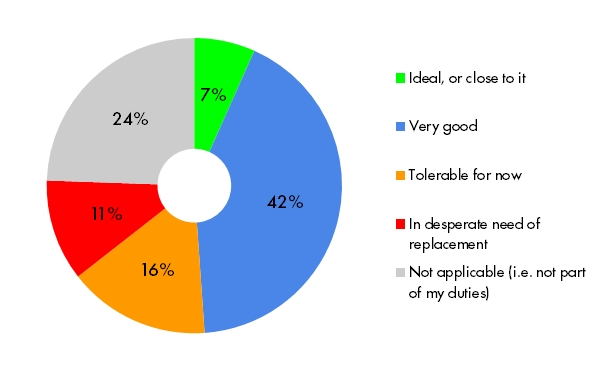
Implementing protocol amendments
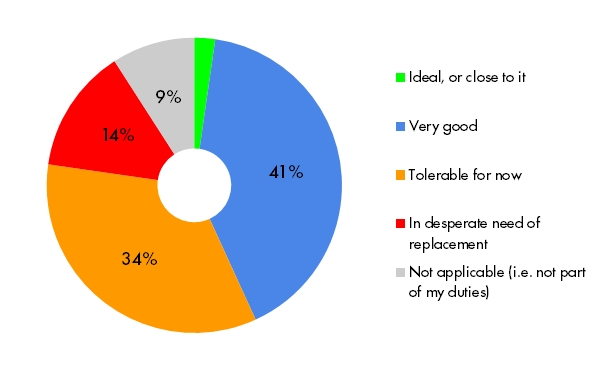
Getting data into SDTM
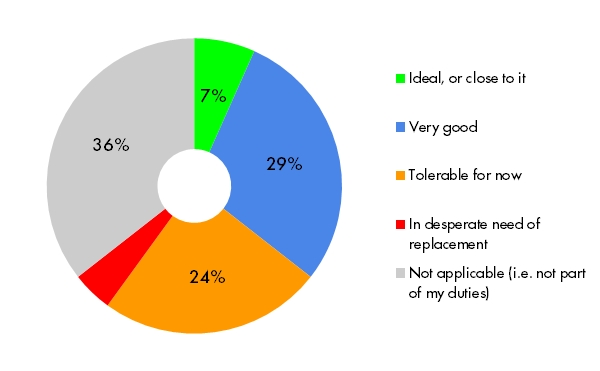
Creating and managing accounts for users and sites
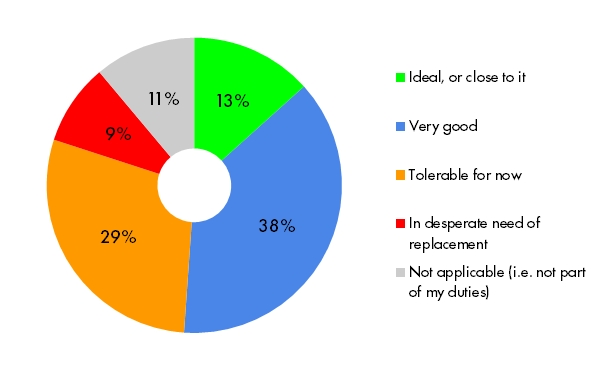
Getting approval and sign-off from stakeholders on study design
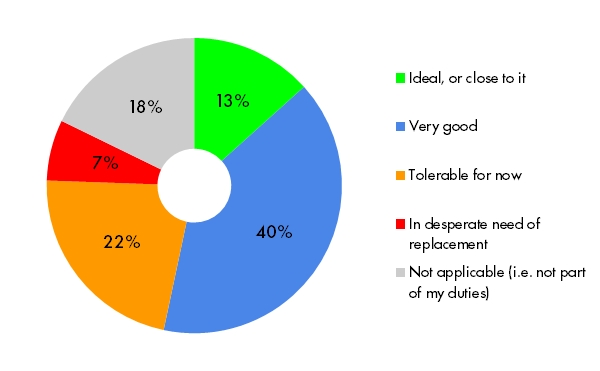
Working with your system’s support team